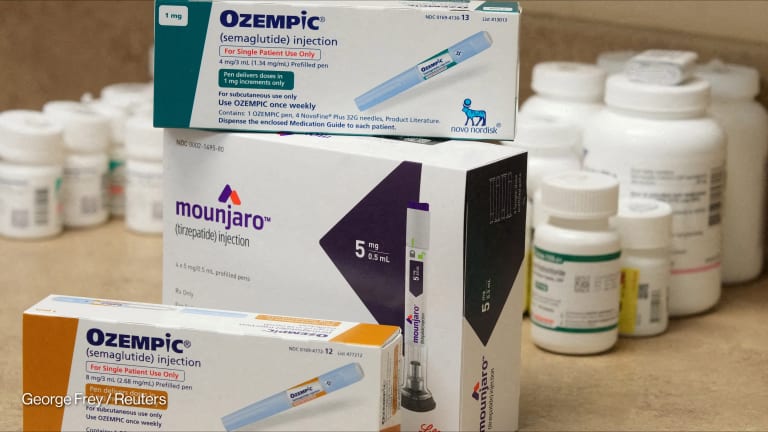
MANILA — Pharmaceutical giant Gilead’s 2018 announcement that it would introduce generic versions of its Harvoni and Epclusa drugs in the United States means patients will have new, cheaper hepatitis C drug options starting this year. But it raises the question: Why not just lower the price of the original drugs?
“The U.S. has, on many levels, a faulty health care and a faulty patent system. And yet that model — that broken model — is being exported to all of these other countries.”
— Priti Krishtel, co-founder, I-MAKHarvoni is currently list priced at upward of $80,000 for an 8-week treatment regimen. With discounts, such as those that come with Medicare, net price comes down to $25,000 — almost the same amount as the reported list price of the generic. The decision to introduce generic versions of the drug is based on the company’s belief that “it is the fastest way to lower list prices for our HCV [hepatitis C] cures without significant disruption to the healthcare system and our business,” said Gilead CEO John Milligan in a statement.
But the $24,000 list price is still “far too high” for most Americans, said Tahir Amin, co-founder of the Initiative for Medicines, Access, and Knowledge, a nonprofit known to challenge unfair drug patents to increase people’s access to affordable medicines. Eighty-two percent of Americans who are aware of having the hepatitis C virus are not getting treatment, he said, adding that cost likely plays a huge factor in that as many of those with the disease come from poor communities.
In India — a leading producer of generic drugs and where Western drug manufacturers often face patent challenges — a generic version of Harvoni retails at less than $1,000 for a 12-week course of treatment, according to I-MAK.
“I don't see the price of $24,000 changing these statistics,” he told Devex. “It is a sad situation where we have branded companies now treating medicines like luxury brands. It allows them to charge astronomical prices at launch and then they offer a slightly cheaper version of the product akin to a subbrand.”
The issue of exorbitant drug pricing has received increased attention in recent years. Governments in low- and middle-income countries often find themselves caught between the need to make available affordable life-saving medicines to their citizens, and upholding drug patents that can run for a few years to decades. This leaves little room for competition and therefore limited treatment options.
But the problem is no longer limited to low- and middle-income countries. In recent years, civil society has shed light on how high drug pricing has also become a burden for those living in high-income countries in Europe and the U.S. AbbVie’s Humira, an anti-inflammatory injectable medication most commonly known to reduce arthritis, currently has a reported average retail price of over $5,000 — covering two injectable pens, the amount typically used in one month — in the U.S., for example.
Humira holds the title of the world’s best-selling prescription drug, and serves as the biggest source of revenue for AbbVie, which I-MAK named and shamed as the worst patent offender in its “Overpatented, Overpriced” report released last year. AbbVie issued 247 patent applications for Humira since receiving U.S. Food and Drug Administration approval in 2002, as a means to keep competition out. Humira remains with no generic competition in the U.S. market.
Bringing the access to medicines debate to the US
For years, I-MAK has focused on fighting back against drug patents in low- and middle-income countries as a result of the pressure they are receiving from their free-trade agreements with Europe and the U.S.
The steep cost of crucial or even lifesaving drugs in the U.S., however, has seen I-MAK expand its work there over the past two years. This includes focusing on challenging drug patents, carrying out evidence-based research, and mobilizing communities — activities the organization has been carrying out for the past 15 years in over 40 low- and middle-income countries. Different organizations, including donor foundations, have reached out to the nonprofit to try and bring its strategy to the U.S.
By coming to the U.S., the organization may be able to raise awareness on the debilitating links that exist between free trade agreements and patent systems, and health and equity, and how drug monopolies are affecting people in large parts of the world, said Priti Krishtel, I-MAK co-founder.
“The U.S. has, on many levels, a faulty health care and a faulty patent system. And yet that model — that broken model — is being exported to all of these other countries. And countries are always having to push back as we saw when we lived in India,” she said. “[The U.S. system] needs a redesign, and maybe then we can be successful in what is being pushed on other countries.”
She knew the move wouldn’t be easy, even with I-MAK’s more than a decade of experience and history of success in challenging drug patents. They tried filing 10 cases against sofosbuvir in 2017, the primary component in Gilead’s hepatitis C drugs, but none of it even reached U.S. court.
“We realized we’re trying to reverse 30 to 40 years of how the pharma companies have formed [in the U.S.], and it’s just going to take time,” Krishtel said.
Instead of filing patent challenges, I-MAK plans to concentrate in the next few years increasing public participation in the patent system in the U.S. Many communities, such as senior citizens, women, the LGBTQ community, and incarcerated populations are hard-hit by the drug pricing crisis, and yet none of them are being included in the conversation.
The organization also hopes to pave the way for more open dialogue with the industry.
“The way the patent law currently stands, I don't know that there's a whole lot of room for compromise either between different industries or between the patient perspective and the pharma perspective … But I think what everybody wants, we can agree on, right? We all want to incentivize new drugs to come to market. We all want to make sure people are actually getting the medicines they need. To do that, we're actually going to have to start a conversation about how do we create a system or redesign to appropriately align incentives with outcome,” Krishtel said.
“If we are able to get slowly some people to start to agree to even be in that conversation, I think it's a huge step forward,” she added.
Update, Jan. 14, 2019: This story has been updated to clarify that I-MAK files patent challenges.