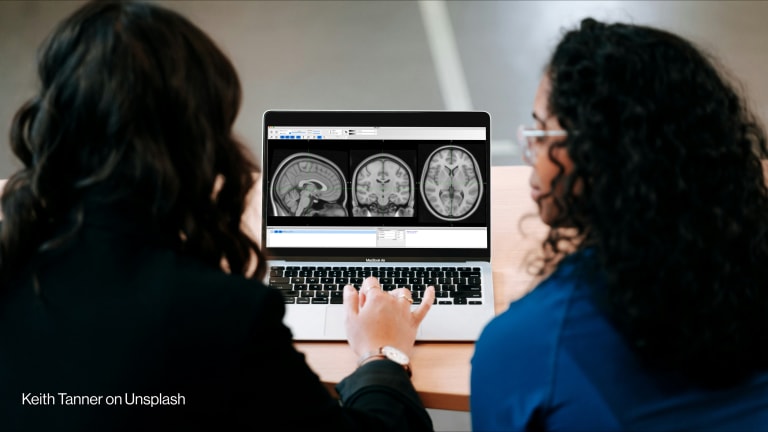
Health monitoring systems are an important fight in the battle to identify, contain and monitor diseases that have the potential to infect thousands, if not millions, of people worldwide. Outbreaks of Ebola and Zika have shown the fundamental flaws in our systems to respond quickly in order to halt a disease in its track — particularly in developing countries.
In 2012, SystemOne was established in Boston to fill this gap in providing fast access to diagnostic data, monitoring and evaluation systems, and connectivity solutions using software and services supporting multiple diagnostic devices and multiple diseases.
“There were definitely periods during the first three years where we had a certain amount of success and we saw the value of it, but we weren’t able to convince or show anyone else the value in the way it could be recognized widely,” Chris Macek, CEO of SystemOne, told Devex. “We definitely considered hanging it up.”
But they stuck with it and now SystemOne is working with partners, including the U.S. Agency for International Development, the World Health Organization and the newly formed Africa Centres for Disease Control and Prevention, and operating in 40 countries within Africa, Central America, the Middle East and Southeast Asia. The newfound demand, which has grown organically without aggressive marketing, has seen SystemOne increase from the four founders to a staff of 26, with a new office opening in Johannesburg.
Devex spoke with Macek about their journey, plans and the road ahead for improved diagnostics in developing countries. Our conversation has been edited for length and clarity.
Can you discuss how SystemOne first came about, and the journey to demonstrating your value to the health care sector in developing countries?
For the first three years of SystemOne we were moving slowly — it was basically proof of concept.
Nigeria was the first country we piloted in and cut our teeth on.
In 2012, the four founders came together and decided to form a new company. A new opportunity arose in Nigeria where there was a particular machine, the first diagnostic device we found, that was suitable for connecting the internet to a database. It was typically used around the world to diagnose tuberculosis.
We worked with Nigeria to first connect a small pilot, and then this was rolled out nationally in a couple of stages over the three-year period. In the end, we connected all of their gene experts to systems for rapid diagnosis and analysis of tuberculosis.
Within a year, we started publishing and publicizing the results of that work and the software we created. A number of other countries started approaching us to see if they could use it to similarly connect their gene experts to diagnose a range of diseases.
Within two years, our product was operating in 10 countries, and by the third year, this number tripled.
We saw different ways of delivering value from connected diagnostics to different audiences, including the health system, logistics and supply chain operators, device manufacturers interested in performance and errors of their devices, and global funders interested in impact and return on investment.
As it has been a slow and organic growth process, how have your range of partnerships come about and what do these allow you to do in terms of improving services and impact?
Our partnership with Daktari Diagnostics was an interesting one. They are a diagnostic device manufacturer, and recognized the importance of connecting their device to allow results to feed into a larger system, including national health systems.
They spent a good deal of time and resources developing their own software to receive results and monitor devices — they probably spent $1 million, which is not a small amount for a start-up company. The problem is that each manufacturer will have systems for their own devices. This is a huge problem for a health department that has to manage the outputs — you can’t understand the face of a disease in your country if it is set across proprietary systems.
“If you connect the diagnostic machines across the continent of Africa, you can do local functions including diagnosis and treatment.”
— Chris Macek, CEO of SystemOneDaktari saw the writing on the wall, and partnered with us to ensure version two of our system, currently in development and due later this year, it is built with their device in mind.
But we are also working with donor countries and multilaterals to support their needs. We have received a challenge grant from USAID to support global disease surveillance for Zika, Ebola and other possible future threats. We are similarly working with the Africa CDC to see if we can support or design a system that would provide disease intelligence from the point of view of the diagnostics. If you connect the diagnostic machines across the continent of Africa, you can do local functions including diagnosis and treatment. But this information can also filter above beyond national borders to the Africa CDC, who has a role in monitoring disease outbreak and other threats across the continent.
What about collaborations with NGOs — are there any interesting collaborations in this space?
Absolutely! In Malawi, we are working with an NGO called Partners in Hope. They run a large hospital in Lilongwe, have 10 centralized labs and are the largest provider of HIV viral load testing and treatment for the country.
They have hired us to come in and solve what is a fairly large problem — right now there are about 700 remote clinics in Malawi where most of the population comes in for tests or treatment. The model they have uses a motorcycle transport system to transport samples and results between clinics weekly. Results can take up to two months to get to the patient, who may also need to walk one or two days to get to their nearest clinic. The time delay is not just an inconvenience: When it is finally received it is irrelevant in terms of clinical treatment.
See related topics:
► Pandemic response a cycle of 'panic and neglect,' says World Bank president
► Why invest in health evidence? Q&A with Chris Murray of IHME
► 3 ways gender data could go 'big'
► Opinion: The World Bank's leadership role in open data — are they back?
What we are doing is installing our system in a central lab, with a server connecting all ten central labs across the country. The real challenge is getting the results to the clinic, where there is no real electricity or available internet access, so we have designed a device that is solar powered, remotely connected to the internet, does not require moving parts and connected to a central network where it searches for and downloads results daily.
We have metrics of the sites activity, and if we are identifying that a clinic is not receiving or acknowledging results, we can call them up to ensure the system is operating correctly and provide service and assistance as required.
15 devices are currently being tested out in the field and we are moving ahead to modify the device to make it even more robust for the African environment. We are anticipating that the end device will costs approximately $2,000, which includes shipping, fees and installation of solar panels associated with the system.
In terms of meeting the diagnostic, data and monitoring requirements of developing countries, monitoring agencies and donors, what capability do your products bring and how will this be improved with future version?
“We clearly recognize the value of connecting devices and streaming data to the health system. As more and more countries, programs, governments and global funders recognize that value, there will be a shift in the funding stream to prioritize this kind of connectivity.”
— Chris Macek, CEO of SystemOneThe first version we built four years ago focused on a single instrument and a single disease — tuberculosis. After we proved the concept, we realized it wouldn’t be too difficult to attach other devices and expand to other disease areas, and we became a multi-disease, multi-device platform.
We realized our first version had limitations in our thinking and architecture. About a year and-a-half ago it became apparent we really needed to begin building again from the ground up with a multi-disease, multi-device platform that could be used very generally.
The other area that we notice that there is challenge with the access to the data — who was able to own the data, who was able to share the data and how that was transmitted and disseminated. One of the basic functions of our platforms is to receive from the diagnostic machine in real time, and to route that information wherever it is appropriate to as many places as necessary, typically determined by the program or operator setting up the system.
With new ideas about disease surveillance and donors looking for greater transparency there are other ways we are establishing the system so it can be shared in appropriate ways, while still maintaining the privacy of patients — detailed information should only be needed at the treatment level.
At the moment we are refining both our software and connectivity platform so it is easier to install, requires less training and is more of a turnkey solution. That is not how it is today. But in terms of being able to scale up, we clearly recognize the value of connecting devices and streaming data to the health system. As more and more countries, programs, governments and global funders recognize that value, there will be a shift in the funding stream to prioritize this kind of connectivity and reporting to offer transparency into the system. Most importantly, wider use of these systems will save lives in developing countries.
Read more international development news online, and subscribe to The Development Newswire to receive the latest from the world’s leading donors and decision-makers — emailed to you free every business day.